The high tech “Nanolek” plant is located in a picturesque forest in the Kirov region near the Levintsy village, 33 kilometers from the capital of the region — Kirov.
Here we produce import — substituting and innovative medicines for the prevention and treatment of socially significant diseases.

Warehouse
Scientific and Practical Center
for Pharmaceutical Development
Administrative
Building and Laboratory
Production of solid
dosage forms
Full-cycle vaccine
manufacturing
30,000 m² factory area
Plant production capacity (per year)
10
million
pre-filled syringes
8
million
vials (solutions, suspensions)
1
million
vials (freeze-dried products)
2
billion
tablets (solid dosage forms)
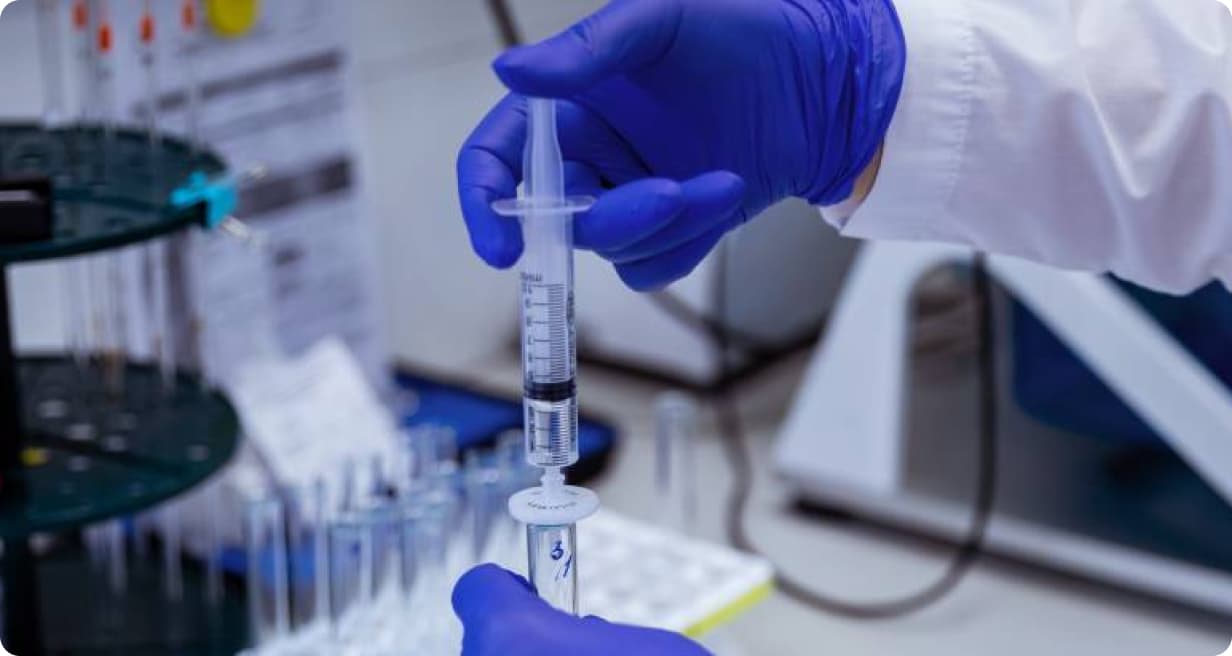
Biotechnological production (BTP)
Launched in 2016. It is the bottling of biotechnological preparations in pre—filled syringes and vials. Full-cycle production, including vaccine substance manufacturing, was launched in 2025.
Solid dosage forms (SLF)
Since 2014, a full cycle of production of solid dosage forms — tablets and capsules — has been introduced.
Quality control
The Holy grail — a laboratory complex where medicines undergo a multi-stage quality control. In particular, there are three laboratories — chemical-biological, microbiological and biochemical.
Certificates
The production of medicines at “Nanolek” complies with international GMP and ISO standards and is regularly certified in other important parameters.
The presence of abundant certification confirms the highest level of quality management in pharmaceutical production and gives preferences when declaring medicines
License for the production of medicinal products
Certificate GOST R ISO 45001-2020
License for Microbiology Laboratory
Certificate GOST R ISO 14001-2016
License for the circulation of narcotic drugs and their precursors
Pharmaceutical activity license
Conclusion on the compliance of the manufacturer of medicinal products with GMP Rules